- Home
- Products
- Water Testing
- pH Paper, Indicator Paper & Test Strips
- Qualitative & Quantitative Test Strips
- Quantofix quaternary ammonium compounds test kit, pack of 100 tests,

- £50.80 excl. VAT
- £60.96incl. VAT
For The Semi-Quantitative Determination Of Ions
Rapid
Most of the QUANTOFIXtest strips can be carried out in 10-120 seconds. That makes the application very quick for the end user.
Convenient
All QUANTOFIXtests are ready to use kits. They are precalibrated and contain all necessary equipment and reagents.
Precise
The colour charts are adjusted and checked using certified standard solutions that are directly traceable to primary NIST standards. The user can be sure to receive accurate readings whenever he tests.
A QUANTOFIX test stick consists of a plastic strip of 0.2mm thickness to which, at the lower end, a test paper has been sealed. The size of the test paper area permits a clear comparison with the fields of equal size of the colour scale provided on the plastic box. The length of the test sticks is sufficient to permit easy handling even in cases where aggressive or otherwise noxious solutions are to be tested.
The plug of the vial contains a desiccant for increased shelf life of the sticks.
Applications
Aquaculture
- QUANTOFIXAmmonium
- QUANTOFIXCarbonate Hardness
- QUANTOFIXNitrate/Nitrite
- QUANTOFIXMultistick for aquarium owners
Electroplating
- QUANTOFIXChromate
- QUANTOFIXCopper
- QUANTOFIXCyanide
- QUANTOFIXNickel
- QUANTOFIXNitrate
- QUANTOFIXZinc
Metal workshops
- QUANTOFIXNitrate
- QUANTOFIXNitrite
- QUANTOFIXLubriCheck
Food processing industry
- QUANTOFIXAscorbic Acid
- QUANTOFIXGlucose
- QUANTOFIXCalcium
- QUANTOFIXCarbonate Hardness
- QUANTOFIXChloride
- QUANTOFIXNitrate/Nitrite
- QUANTOFIXPeroxide
- QUANTOFIXPhosphate
- QUANTOFIXQUAT
- QUANTOFIXSulphite
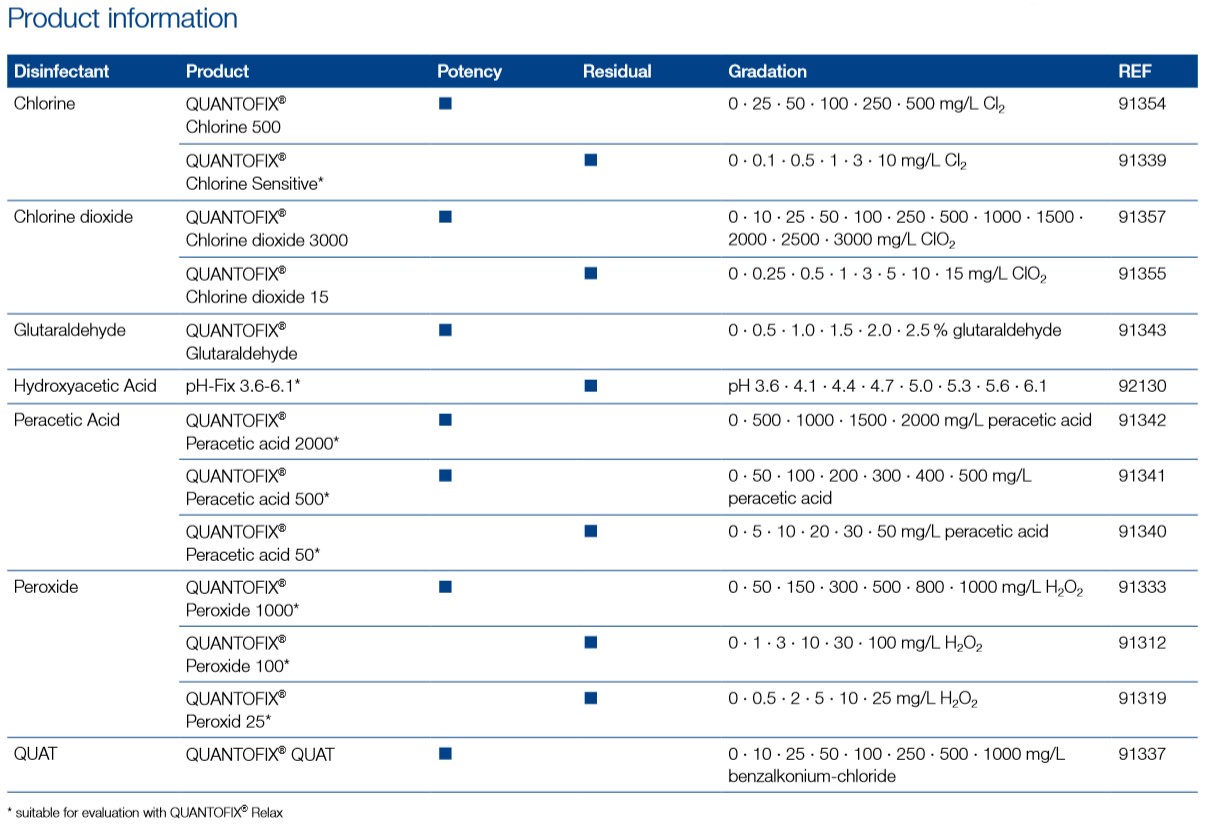
| Platform | Semi-quantitative test strips |
| Analyte | QUAT |
| Brand | QUANTOFIX |
| Gradation | QUAT - 0?•?10?•?25?•?50?•?100?•?250?•?500?•?1000? mg/L Benzalkonium chloride |
| Color change | QUAT - yellow?→?blue-green |
| Evaluable on reflectometer | No |
| Method | QUAT - Mixed indicator |
| Additional reagent | No |
| Storage temperature | 4−30 °C |
| Shelf life | 2.5 Years |
| Scope of delivery | 100 test strips in a tube, instruction leaflet |
| Hazardous material | No |
Disinfectant testing- Rapid Tests for Healthcare
- Quick & easy
- Reliable results
- Higher patient safety
Health care professionals use different types of disinfectants and sanitizers for general disinfection and for cleaning of medical devices. Prior to the disinfection of devices, it proved useful to control the potency of the disinfection baths. After disinfection, all devices have to be rinsed and controlled for sanitizer residues prior to the next use. Dip & Read tests allow the quick and easy detection of sanitizer residues. These test strips increase patient safety while making healthcare professionals’ work easier and more efficient.
The QUANTOFIX® Relax allows the automatic reading of QUANTOFIX® test strips. Just place the strip on the strip adaptor and the Relax will give a precise and reliable result within seconds. The results are printable, saved internally and can be transmitted to a PC.
QUANTOFIX® Relax – for ease, reliable QC procedures
- Highest precision
- Objective results
- Easy result management
QUANTOFIX test strips meet all requirements of a modern rapid test. The colour of the reactive pad changes depending on the concentration of an analyte in the sample. The evaluation is usually carried out visually by a comparison of the reaction colour with a multi-stage colour scale.
Rapid
- Just Dip & Read
- Results within seconds
- Always ready for use
Easy
- No calibration
- No maintenance
- No accessories
Reliable
- Desiccant in the stopper for optimal protection of the strips against humidity
- Colour chart confirmed with traceable standards
- Automatic evaluation with QUANTOFIX Relax for safe documentation
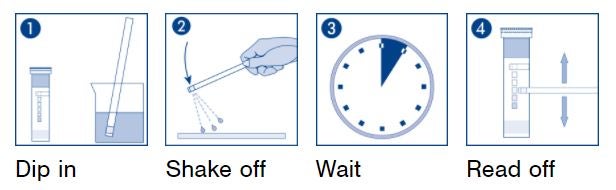
Application of QUANTOFIX test strips
Easy analysis directly at the point of interest
Analytical professionals as well as occasional testers appreciate QUANTOFIX test strips for the fast and easy analysis directly at the point of interest. Often, these tests are used to quickly check whether important parameters are in the desired range. They deliver an immediate result and thus enable a fast response.
Complete mini-lab
QUANTOFIX tests are immediately ready‑to‑use. They do not require additional accessories.
The test strips are intended for single use, maintenance or calibration are not required.
CE-mark for medical applications
Some QUANTOFIX test strips are tested and approved for medical applications and carry a CE-mark for medical products 93/42/EWG. They meet the special demands of health care professionals and ensure safe results in the medical field.
Quantitative, documented results with QUANTOFIX Relax
The strip reader QUANTOFIX Relax provides objective and quantitative results for many important parameters. Measurement data including time, date and sample ID are printed, stored and can be transmitted to an information system. This allows the rapid and reliable documentation of test results, which proved to be especially useful for QC departments.